Though many of us would like to deny it, our neighbors affect us. Their loud music disturbs our sleep. The scent of their dinner wafts through an open window. The sight of their new puppy brings a smile. We interact with our environment every moment of the day. And so do lung cancer cells.
A Brief History
Traditionally, we have studied tumor cells in isolation. We often study single cells in a petri dish, or under a microscope, to understand how they work. This has been very important and effective. Studying individual cells continues in labs around the world and it leads us to understand many fundamentals of tumor biology including the intricacies of DNA, RNA, and protein.
However, for over a century, researchers have also been learning about complicated relationships between cells. Back in 1907, a scientist first showed that individual cells from a sea sponge could spontaneously reorganize themselves into a sponge formation.
Since then, we’ve made steady progress in understanding relationships between cells. Eighty years later, in 1987, scientists had improved our understanding of the cellular environment so much that they used petri dishes to grow epithelial breast cells that formed milk ducts and produced milk.
By the 1990s, stem cell research had gained momentum, and researchers were learning how to coax stem cells to grow into a variety of cell types.
Organoids are Here
Researchers have taken the power of stem cells and combined it with our knowledge of organ development to create organoids: stem cells grown to form 3D models of a specific organ or tissue that can be used for research.
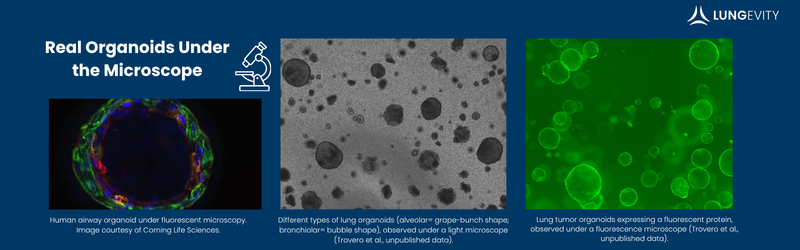
These “mini-organs” don’t look like much to the human eye, but under a microscope you can see they are an organized system of cells with the potential to teach us about organs and tissues within the human body.
While there are many emerging technologies for how organoids are created and maintained, most methods still require preventing the organoids from touching the plastic surface of the petri dish to retain their 3D shape.
Some researchers use organoids that are grown on a special base layer which mimics the extracellular space in the human body, while others grow organoids in a droplet of nutrition.
Organoids are an exciting addition to research laboratories because they offer a 3D model to study cells within their complex environments. Today’s organoids can be developed using patient tissue samples for many different types of research, including:
- Studying ways to regenerate organs
- Testing the toxicity of consumer products
- Identifying potential treatments for disease
Organoids in Lung Cancer
A breakthrough research study, published in Nature Communications in 2019, showcased the potential to use patient-derived lung cancer organoids to look for new treatments. This published data also showed that organoids are strong models for studying several different subtypes of lung cancer, such as EGFR-positive, ALK-positive or BRCA2-positive non-small cell lung cancer (NSCLC).
That same year, LUNGevity leveraged the scientific potential of this new technology by partnering with the Broad Institute of MIT and Harvard to build a library of lung cancer- focused organoids. This collaboration empowered lung cancer patients by offering a way to donate tissue samples for creating organoid libraries and speeding progress in lung cancer research.
Today, organoid models are being used to study many aspects of lung cancer.
In 2023, LUNGevity awarded a research grant to Maria F. Trovero, PhD, a postdoctoral fellow at Boston Children’s Hospital, to study the role of a protein, METTL3, in lung cancer. This work uses organoids that model KRAS-positive NSCLC to identify a biomarker for early detection of the disease and to improve options for treating advanced lung cancer.
During her project, Dr. Trovero found that METTL3 has different roles that change based on which types of cells are nearby in the tumor microenvironment. Her findings underscore the importance of analyzing both tumor cells and their surroundings when developing optimized treatments for patients.
“Organoids are a clever bridge between the lab and clinical trials,” notes Dr. Trovero. “These 3D models offer a safe and effective way to study the effects of lung cancer treatments on tumors before we start in vivo experiments and clinical trials.”
Organoids are also being used in other areas of lung cancer research, such as improving immune cell penetration during immunotherapy, enhancing personalized medicine, and understanding the 3D structural changes that occur during lung cancer treatment.
Organoids have become an important tool for researchers to study complex relationships between cells within organs and tumors. They are reshaping how researchers study lung cancer and bringing us closer to a world where no one dies from the disease.
Additional reading:

